UV visible Spectrophotometer Setup and Beer-Lambert Law
In this article we are going to have a brief discussion of UV visible spectroscopy and the Beer-Lambert law.
UV vis Spectrophotometer Setup
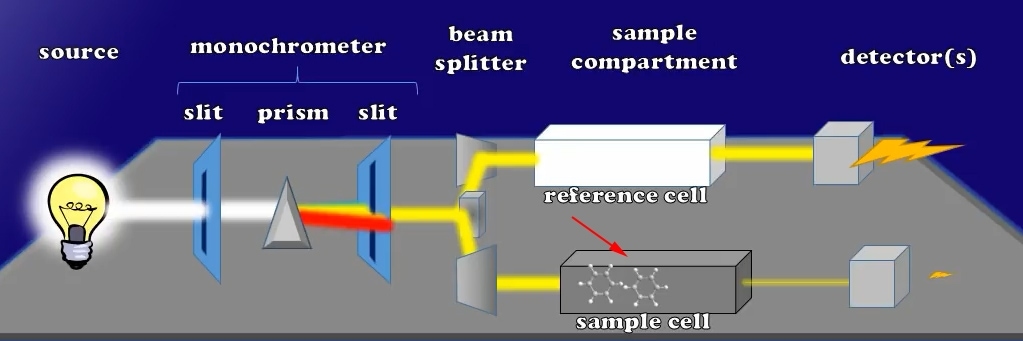
To start with our session below is an engineering design that is a relatively typical type of setup for a UV visible spectrophotometer. A variety of different brands and designs are available to manufacture a spectrophotometer but this is among the simplest ways. Let’s continue from here.

Simple UV vis Spectrophotometer Setup
Simple Spectrophotometer Parts
- The first component is a source lamp that can be as basic as a motor scooter headlamp or a more complex Deuterium lamp or even xenon arc lamp style.
- The next part is a monochromator, which consists of two slits separated by a prism or a grid of diffraction. We’ll talk about the use of slits below.
- The next device of this spectrophotometer is the so-called beam splitter which divides the light beam into two parallel light beams.This part consists of 2 prisms.
- The next component is the sample compartment where the reference and sample cuvettes locate.
- And the final element is the detectors which are sensors that can be monitored by a computer to turn the impact of photons into electric current.

5 Parts of Spectrophotometer Design
So now that we have each of the smaller elements inside our simple spectrophotometer identified. Let’s turn on the instrument, and see what’s going to happen.
Let’s light the lamp light source creating different lights in a range of wavelengths. This light passes through this first monochromator slit, which makes sure that all the light photons will travel along parallel pathways; later when they reach the prism, the lights will be able to refract into a color rainbow.
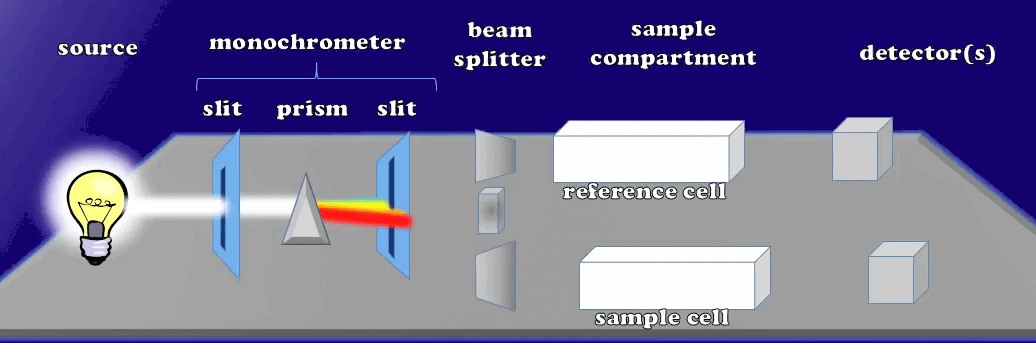
Lights Travel Parallel Pathways after Monochromator
Here each wavelength of light moves to the other slit, so that only a preset wavelength of light is able to pass through. In the monochromator the second slit reaches the beam splitter which makes the light into two beams of equal intensity. These two equal intensity beams will, pass through the sample compartment of different cuvettes: the reference and sample cuvettes.
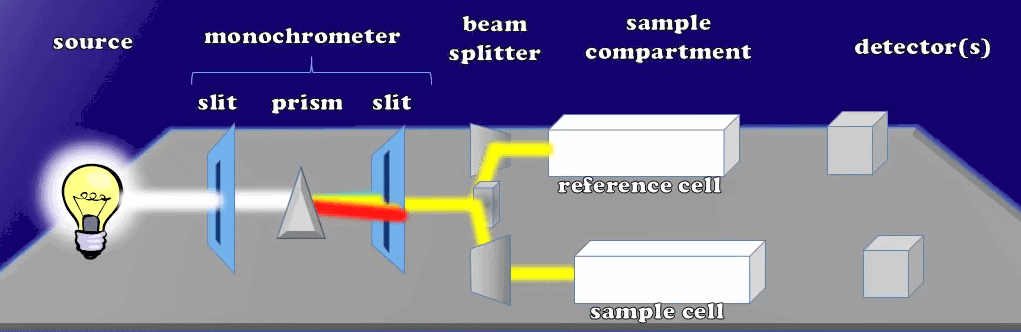
Beams Traverse Each Cuvette with Equal Intensity
While the beam exits these cuvettes and hits the detection system that fires away to create an electric current, you will find that both the reference and sample cuvettes have the same light intensity at this moment, as a result the current generated by each detector is the same.

Identical Exit Current for Both Cuvettes
Spectrophotometer Data Consequences
So both the current intensity detected by each of the two detectors is identical in this case. If we take into account the ratio of the intensity that exits the sample cuvette to that which leaves the reference cuvette, we can see that our sample cuvette transmission is 100% that of the reference cuvette. We will, therefore, map this 100% transmission here at zero concentration.

I=Sample Cuvette I0=Reference Cuvette
Let us now add a little sample to that cuvette, which could absorb the light. Note that the intensity of the light from the sample cuvette decreases when I do this. There is therefore also a reduction of the current generated by its detector, respectively.
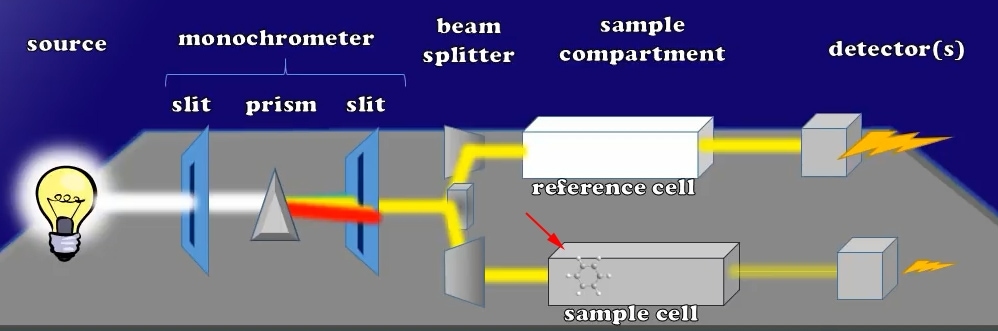
Current Decrease after Sample Added
As such, the intensity ratio is not 100% anymore, in this case let’s say it goes down to 50% at a given concentration X.
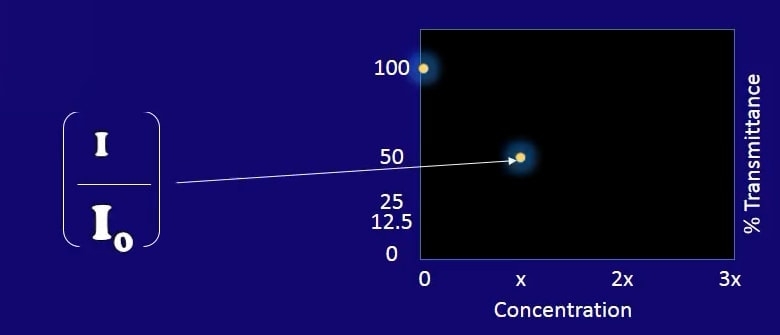
Intensity Ratio Goes Down to 50%
If I add another equivalent sample to the sample cuvette that further reduces the current strength. The sample reduces the intensity of light passing through the sample cuvette by just one-half for each equivalent sample concentration we add.
Second Sample Added
Now let’s calculate the new current intensities of the sample and reference cuvettes, we’ll notice that it’s 25% transmittance and an incremental increase in the concentration at the same time.

I=Sample Cuvette I0=Reference Cuvette
Add sample one more time the sample cuvette leads to another decrease by 50% and consequently a percent transmittance of 12.5%. The concentration is now 3x.

I=Sample Cuvette I0=Reference Cuvette
We now have enough data to see something really interesting.
The relationship between the transmission percent and the sample concentration is not linear. It’s exponential instead.
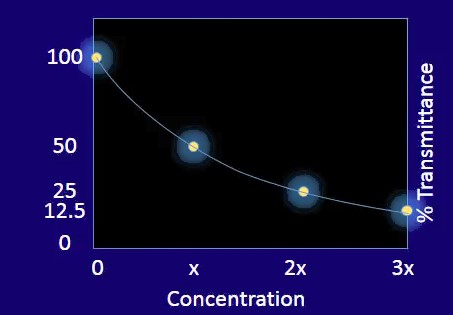
While this data is very useful, researchers and spectroscopists prefer to analyze linear relationships within the data results, as long as it is possible, because this will make the discussion much clearer and it is far easier to forecast how things will be if we have a straightforward linear plot.
Hello Beer-Lambert Law
That is where Beer-Lambert law becomes useful. You might have an idea that now we have an exponential connection between the transmission percentage and my concentration.
In this equation, we can see that the concentration is a term in the exponent (conc).

August Beer converted the transmittance percentage to a new unit called absorbance. By taking the logarithm or in really the negative logarithm of the transmittance we have the following equation:

By using the logarithm of an exponential function a linear function is created. The data are therefore considerably easier to look at when plotted as absorbance rather than transmittance. Not to mention that by extrapolating or interpolating within the dataset we’ve collected, it is much easier to predict the exact absorption.

This is why we convert percentage transmittance into absorbance so often by performing UV visible spectroscopy. This is the application of Beer-Lambert law.
That’s all for now! Thank you for your time reading!
Presenter: Professor Davis from ChemSurvival
Link to video: https://www.youtube.com/watch?v=wxrAELeXlek
